Shining a Light on Mobile Device Virus Control Using drylin® Linear Slides

For nearly three decades, ultraviolet light has been used as a source of disinfectant, particularly in the medical community. But as the team at CleanSlate UV discovered in creating a device to eliminate bacteria on cell phones and other mobile devices, using it as a disinfectant requires extreme care, testing and precision.
The CleanSlate UV Sanitizer can eradicate 99.9998 percent of MRSA (Methicillin-resistant Staphylococcus aureus) in 20 seconds. The device works on smartphones, tablets, badges and other portable items, does not require any training to use and disinfects without harsh chemicals that can damage electronics.
Demand for sanitization of mobile devices is critical. Studies have found that 94 percent of cell phones used by hospital staff bore contaminants. In another report, 89 medical workplace employees were aware mobile phones could be a source of contamination, but only 13 disinfected their phones regularly.
“More and more, mobile devices are being used in healthcare in the context of patient care,’’ said Josée Shymanski, Manager of Infection Control at Montfort Hospital in Ottawa. “For example, we can use these devices for patient teaching, for patients to complete questionnaires or surveys, and to access information on the web. In addition, pretty much everyone has a cellular phone. We know that these devices will become contaminated with germs over time, sometimes with germs that can be harmful. We do not want these devices to become a source of infection for our patients and staff.”
The use of ultraviolet light, however, requires extreme care. Prolonged exposure to ultraviolet light can impact skin, eyes and the immune system. The CleanSlate team conducted extensive research to identify the list of pathogens that are most commonly found in healthcare facilities causing healthcare-acquired infections. The device they created deactivates bacteria and spores and keeps users safe from harmful UV rays. It was a laborious, extensive and delicate balancing act.
“We have taken every possible use into consideration to ensure the personnel is not exposed to UV-C light under normal use and maintenance,’’ said Manju Anand, Chief Technology Officer for CleanSlate UV.
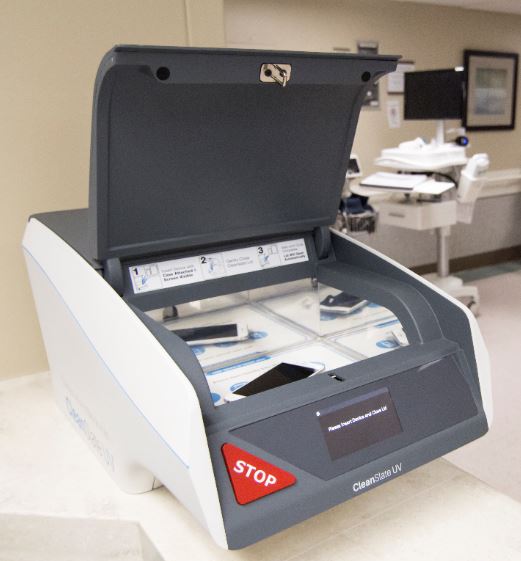
How the device works
The user deposits the mobile device into the machine, closes a lid and waits 20 seconds for the device to be cleaned. Once completed, the lid opens automatically and the mobile device can then be removed with clean hands. The device can sanitize multiple items at once, and includes RFID-enabled tracking and compliance auditing. The UV-C light does not dry out or degrade materials, which occur with chemical wipes.
Mobile devices are placed on a movable chamber that transports the device into a UV chamber, where the sanitization occurs. After 20 seconds, the device slides out and is ready for use.
The device utilizes short-wavelength ultraviolet light (UV-C), which destroys nucleic acids and breaks apart germ DNA, preventing them from being able to function or reproduce. The light does not physically remove the cell, but causes damage to the nucleic acid of microorganisms, preventing the DNA from being unzipped for replication. The organism is unable to reproduce; when it tries to replicate, the organism dies.
Safe use of UV light
In CleanSlate’s early stages, the team conducted extensive research within the hospital network and research publications. “With the help of research publications, we identified the minimum required dosage to achieve the desired kill rates for the selected pathogens. The hardest one was Clostridioides difficile (also commonly known as C.diff),’’ Anand said.
CleanSlate developed a UV test chamber to adjust light source, intensity, material and finish of the chamber, and duration of exposure. “Using a radiometer, we measured the intensity and uniformity of UV light throughout the chamber,’’ Anand said. “Through a number of tests, the best combination of the UV source, custom coatings for the chamber surface and the size of the chamber was finalized.”
The UV test chamber was then sent to a third party test lab to conduct efficacy testing according to ASTM E1153 which is the current standard test method for sanitizers recommended for hard, non-porous surfaces.
Tests were conducted at several exposures to measure UV-C dosage (intensity x duration). These test results were the foundation for CleanSlate’s commercial version design and development.
“We conducted extensive research on devices that require frequent sanitization, which defines our chamber size and duration of sanitization in healthcare settings without disrupting the workflow,’’ Anand said. “With the help of an industrial design firm, we finalized the UI/UX and look of the product so that it integrates well into healthcare facilities without disrupting the workflow, looks like modern medical device and is easy and intuitive to use without any training.”
Teams designing the chamber faced multiple challenges in maintaining the proper UV exposure. “With the help of a thermal simulation tool, we optimized the chamber design further to enhance the airflow within the chamber and temperature sensing technology was integrated which monitors the temperature all the time,’’ Anand said. “Any point the temperature is outside the set limits, it warns the user and stops from anyone using the device by going to servicing mode.”
Chamber technology
A critical component of the product is a sliding chamber that is lubrication- and maintenance-free. The linear guide is made by igus®, a Germany-based manufacturer of motion plastics products. The company runs its North American operations out of Providence, R.I. The guides slide, instead of roll, and are cost-effective and highly flexible. Drylin W guides are resistant to dirt and dust due to dry operation, and are typically used in medical equipment and devices, packaging machines, furniture and robotics.
 “During the initial R&D stage, we discovered that the UV lights must be on and warm for effective sanitization in 20 seconds,’’ said Kevin Wright, the Canadian sales manager for igus®. “Since the UV light is dangerous to human skin and eye, we had to design a moving chamber that transports the device into the UV chamber when the sanitization was initiated by the user. It was important to have a bearing system that was extremely quiet, especially in the evening shifts where any noise from the devices can be disturbing to the patients and staff.”
“During the initial R&D stage, we discovered that the UV lights must be on and warm for effective sanitization in 20 seconds,’’ said Kevin Wright, the Canadian sales manager for igus®. “Since the UV light is dangerous to human skin and eye, we had to design a moving chamber that transports the device into the UV chamber when the sanitization was initiated by the user. It was important to have a bearing system that was extremely quiet, especially in the evening shifts where any noise from the devices can be disturbing to the patients and staff.”
Anand said the company tried steel bearings, but they fell far short of their expected service life and required lubricants, which could not be used in a healthcare facility. “Reliability was utmost, as any downtime due to CleanSlate device breakdown would result in the ineffective sanitization of mobile devices using chemical wipes, which could damage or degrade the electronic devices used in hospitals,’’ Anand said.
Worldwide use
More than 40 hospital systems, biotech companies and food processors in the United States, Canada, Australia and Hong Kong are using the CleanSlate UV Sanitizer to control the spread of pathogens in their facilities.
“So many people come to a hospital every day; having this type of machine available for them to use reinforces the message that hygiene is important,’’ Shymanski said. “Combining cleaning your phone with cleaning your hands in one hygiene moment that takes 20 seconds is a good practice and shows that as an organization, we value clean care, and hopefully that translates to our patients feeling safe.”
Interested in using hygienic linear bearings within your machine or design? Do not hesitate to contact the Product Manager for drylin® linear technology for igus North America.



